  
Modern understanding of atomic structure has shown that the numbers andĪrrangement of the electrons in the atom are responsible for the periodicity Was found to have the properties which Mendeleev had predicted. Their attitude changed when theĮlement gallium was discovered in 1875 and As is often the case with new ideas, chemists wereĪt first skeptical of this classification. He alsoĭeduced their more important properties from the properties of adjacentĮlements in the table. Not only did Mendeleyev leave gaps in the table for new elements. The elements were arranged in short sequences with similar The form of Mendeleyev's table of 1869 has been followed closely in the Way were able to accommodate elements with similar properties in the same To recognize the possibility of their being further elements still to discover.Īccordingly they left gaps in the table for unknown elements, and in this In much the same way as Newlands had done, but both of them had the foresight They arranged the elements according to their atomic weights, In 1869 the Russian chemist Dmitri Mendeleyev and Julius Lothar Meyer, a German, published somewhat similar periodic tables Scale, and his deduction relating to the periodic repetition of similarĮlements became known as the law of octaves. Newlands likened this repetition to the octaves of the musical Of the first." This, in his table, fluorine is No. That "the eighth element, starting from the given one, is a kind of repetition The known elements in ascending order of their atomic weights and noticed While studying the revised value for the atomic weights, John Newlands,Ī British industrial chemist, realized that there was some correlation betweenĪtomic weights and the chemical properties of the elements. Measurements are now related to the atomic weight of carbon isotope 12 as Standard upon which atomic weights are based has since been modified. Number of times one atom of it is heavier than an atom of hydrogen. The atomic weight of an element used to be taken as the Stas (1813–1891)Īnd others set out in the 1850s to determine afresh the atomic weights (see relative atomic mass) of the It was against this background that Stanislao Cannizzaro, Measurements in an attempt to obtain more accurate and reliable results. As a consequence, joints tended to leak, and results were unreliable.Īs such improved apparatus and more accurate instruments became available,Ĭhemists and workers in other branches of science were not only able toĮxtend their studies but also to consolidate previous knowledge by repeating Thus, before the introduction of rubber tubing in the 1840s connectionsīetween pieces of apparatus could only be made with glass, pottery, metal, The discoveryĪnd development of new materials during the 19th century contributed toīetter scientific apparatus and made possible more accurate experimental Perhaps based on measurements which would yield numerical values. There was, therefore, a need for a more positive means of classification, The resemblance between individuals is only slight. Of some of the families had not been isolated and also because in some groups However, such a classificationĭid not accommodate all the elements. 
The elements in each group have similar properties. Metals (lithium, sodium, and potassium) and the halogens (fluorine, chlorine, bromine, and iodine) were in some way related since Of more elements it became obvious that some of them, notably the alkali In 1800 only 33 chemical elements had been isolated,īut during the next 60 years this total was almost doubled. In the early years of the 19th century some form of classification of theĮlements began to emerge. In a period increases as one moves down the periodic table because as theĮnergy level of the atom increases, the number of energy sub-levels per Level an electron in an atom of that element occupies (in the unexcited state). The period number of an element signifies the highest energy 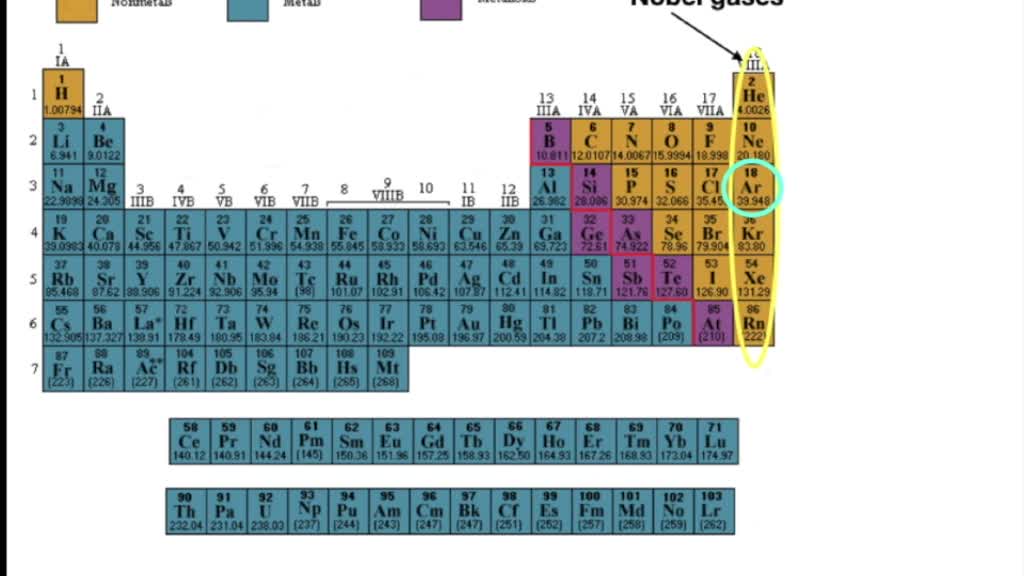
The different rows of elements are called periods. Rows and columns to illustrate periodic similarities and trends in physicalĪnd chemical properties. The periodic table is a table of the elements in order of atomic number, arranged in 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
